VB News Desk: A Potential New COVID Treatment and the Race for a SARS-CoV-2 Vaccine
Posted on 7/3/20 by Laura Snider
As the COVID-19 pandemic continues around the world, the research community has been abuzz with studies centered on finding treatments, and ultimately a vaccine for the virus that causes COVID, SARS-CoV-2.
What they’ve found is that some preexisting drugs, used to treat other conditions, can also be used to help patients with COVID recover more quickly or to decrease the severity of certain COVID symptoms, such as inflammation in the lungs. In addition, there are a variety of vaccines currently being developed and tested—hopefully, one or more of them will show successful results in clinical trials and be able to go to production and distribution soon!
1. Dexamethasone shows life-saving potential in recent trials
Newly-released trial results from researchers in the UK suggest that doctors might be able to add the steroid dexamethasone to their anti-COVID arsenal. Previously, dexamethasone has been used to treat asthma, arthritis, and some skin conditions because it has anti-inflammatory effects. The wide availability and low cost of dexamethasone make it an attractive option, since other drugs being used to treat COVID, like the antiviral Remdesivir, are in much shorter supply.
Preliminary reporting of the study’s results indicates that dexamethasone reduced the death rate by 35% for patients who were on ventilators. It reduced the death rate by 20% for patients who were receiving oxygen but were not ventilated. For patients who did not require oxygen, there was not a significant benefit to treatment with dexamethasone.
Why would an anti-inflammatory help the most severely ill COVID patients? In some cases of COVID, the body’s own immune response to the virus goes into overdrive, causing a “cytokine storm.” When this happens, it can result in acute respiratory distress syndrome (ARDS), in which fluid builds up in the alveoli, leading to low blood oxygen levels, and potentially even death.
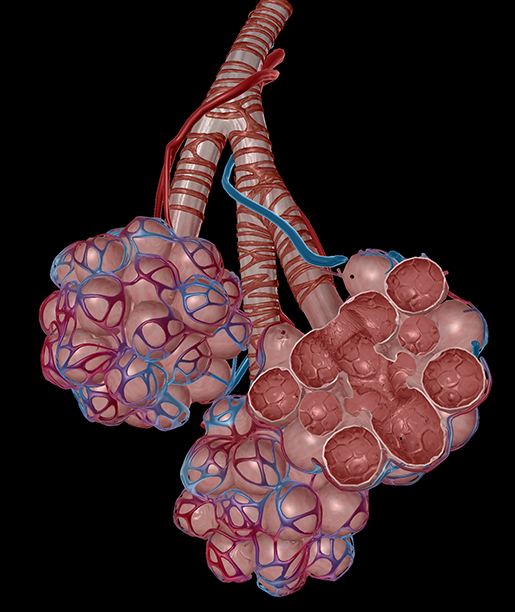 Healthy alveoli are small air sacs where gas exchange takes place. This keeps the blood supplied with fresh oxygen. Image from Human Anatomy Atlas.
Healthy alveoli are small air sacs where gas exchange takes place. This keeps the blood supplied with fresh oxygen. Image from Human Anatomy Atlas.
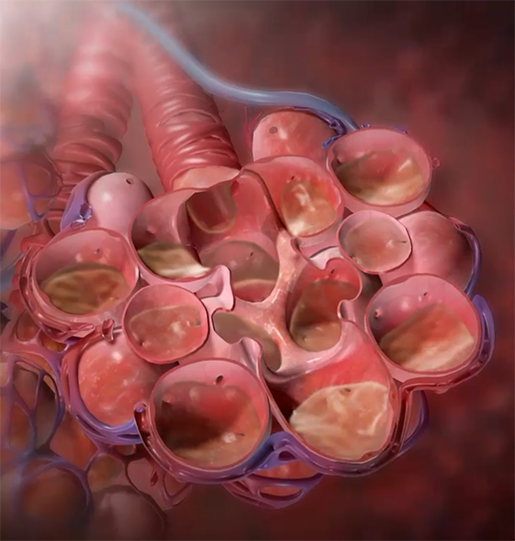 In conditions like acute respiratory distress syndrome (ARDS) and pneumonia, fluid fills the alveoli in the lungs, making gas exchange difficult. Animation screenshot from Physiology & Pathology.
In conditions like acute respiratory distress syndrome (ARDS) and pneumonia, fluid fills the alveoli in the lungs, making gas exchange difficult. Animation screenshot from Physiology & Pathology.
Since the full paper detailing the dexamethasone study hasn’t been released yet, experts like infectious disease specialist Dr. Kristen Lyke from the University of Maryland School of Medicine urge that our optimism remains cautious. Everyone is eager for good news, but it’s important to make sure that we take a close, analytical look at the methodology of studies involving treatments for COVID.
2. Vaccine research is in full swing
While doctors work to treat patients, the pharmaceutical industry has been working towards the development of a COVID vaccine. Many different companies and research institutions have been developing and testing vaccines that help the human body develop immunity to the SARS-CoV-2 virus using several different strategies.
Virus vaccines introduce a weakened or inactivated virus (or part of a virus) to the body. This allows the body’s immune system to develop a response to the virus without an actual infection happening. Protein vaccines use proteins or fragments of proteins from a virus (like the spike proteins of SARS-CoV-2) to train the immune system to recognize and respond to the virus.
Viral vector vaccines use live viruses to carry DNA (from the virus the vaccine is intended to protect against) that codes for specific antigenic proteins into the body’s cells. Nucleic acid vaccines aim to use DNA or RNA from the virus to provoke an immune response—RNA vaccines are very new, and there haven’t yet been any approved for use in humans.
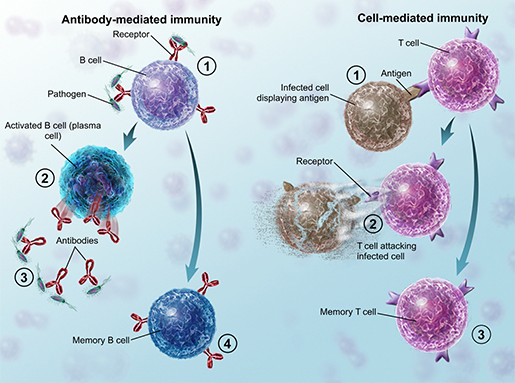 Vaccines help “train” the immune system by mimicking the pathogen illustrated in the antibody-mediated immunity side of the illustration above. Some types of vaccines, like viral vector vaccines and DNA vaccines, interact with cell-mediated immunity. Illustration from Anatomy & Physiology.
Vaccines help “train” the immune system by mimicking the pathogen illustrated in the antibody-mediated immunity side of the illustration above. Some types of vaccines, like viral vector vaccines and DNA vaccines, interact with cell-mediated immunity. Illustration from Anatomy & Physiology.
According to the New York Times Coronavirus Vaccine Tracker (a frequently updated resource that categorizes current vaccine research efforts by vaccine type and clinical trial phase), there are currently over 135 different coronavirus vaccines in development. Most haven’t been tested in humans yet, but two so far are in Phase III clinical trials: a vaccine being developed by Oxford University’s Jenner Institute and a tuberculosis vaccine developed years ago that could provide some protection against SARS-CoV-2.
A Phase III clinical trial largely follows the same format as a Phase II clinical trial, in which a vaccine is given to a large group of people and a placebo is given to another group. Later, the researchers measure how many people from each group have become infected. (No one is deliberately infected with the virus, by the way. Scientists just wait to see how many infections occur.) The main difference between Phase II and Phase III clinical trials is that Phase III trials are meant to confirm data from Phase II and to allow for adjusting the dosage and monitoring side effects.
An increasing number of vaccines are now reaching Phase II clinical trials. This is encouraging news, and means that although we don’t have an approved SARS-CoV-2 vaccine yet, estimates that we will have one by next year likely aren’t too far off.
To learn more about the respiratory system and about immune responses, check out these articles from the Visible Body Learn Site and the Visible Body Blog:
- Learn Site: How the Immune System Protects the Body
- Learn Site: Respiratory System Pathologies
- Blog: Exploring Lung Pathologies with Physiology & Pathology
- Blog: Innate and Adaptive Immunity
If you’re interested in seeing what past pandemics can teach us about our current situation, check out this blog post!
Be sure to subscribe to the Visible Body Blog for more anatomy awesomeness!
Are you an instructor? We have award-winning 3D products and resources for your anatomy and physiology course! Learn more here.



