Feeling Hormonal: A Look at the Endocrine System
Posted on 6/16/17 by Marian Siljeholm
“Feeling hormonal?” Or, “It’s just the hormones!” We all complain about hormones without really knowing where they come from or what they do. Turns out, these chemical messengers don’t deserve as much flack as they’re getting. In fact, we should be worshipping the glands where these chemical messengers secrete from. Vital for survival, hormones provide the relay system every cell needs to send information and instructions through the body. Where do these miraculous messengers come from? The endocrine system: a network of glands throughout the body that regulates some pretty important functions like body temperature, metabolism, growth, and sexual development.
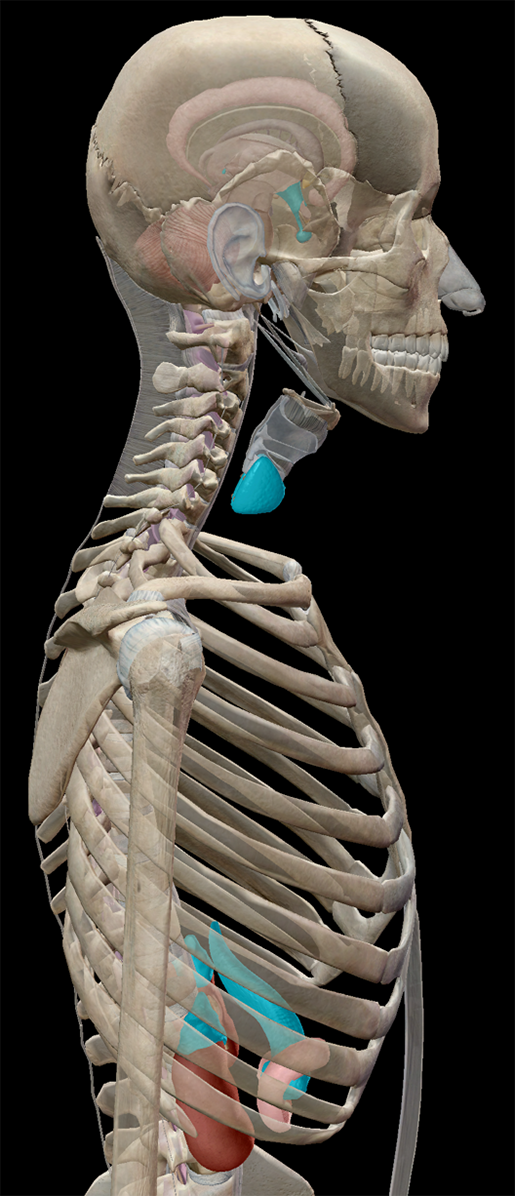 Image from Human Anatomy Atlas.
Image from Human Anatomy Atlas.
Like the digestive system, the endocrine system is made up of primary and secondary organs (we can’t all come in first). The primary organs include the hypothalamus and pancreas along with the pituitary, thyroid, pineal, and parathyroid and adrenal glands, which all release regulatory hormones, while the secondaries include the gonads, kidneys, heart, and thymus.
Together, all the endocrine glands and their hormones regulate things like metabolism and the sleep cycle, as well as growth and development. You don’t have to tell this system twice that there’s no place like homeostasis.
But what is the endocrine system, exactly, and how does this complex, crucial network affect the body? Read on to find out!
Let’s start with hormones, and then move on to their homes. When endocrine glands and organs secrete hormones into the bloodstream, those chemical messengers go everywhere. Our hormones are picky, though. They’ll only bind to specific target cells with receptors just for them. That binding triggers changes in the behavior of the cells. If a type of hormone binds to enough cells, it will adjust the corresponding functions of organs and body processes.
And where does it start? The brain (typical). Two hormone command centers in the brain—namely, the hypothalamus and the pituitary gland—are the dynamic duo in charge of this entire operation. After receiving signals from other brain regions, the hypothalamus translates them into the endocrine language: hormones, which then travel to the pituitary. This signal sets in motion signals to either inhibit or release certain hormones, some of which will act directly, affecting skeletal and muscular development and processes such as birth and nurturing, or indirectly, serving as commands to subordinate endocrine glands.
Hypothalamus
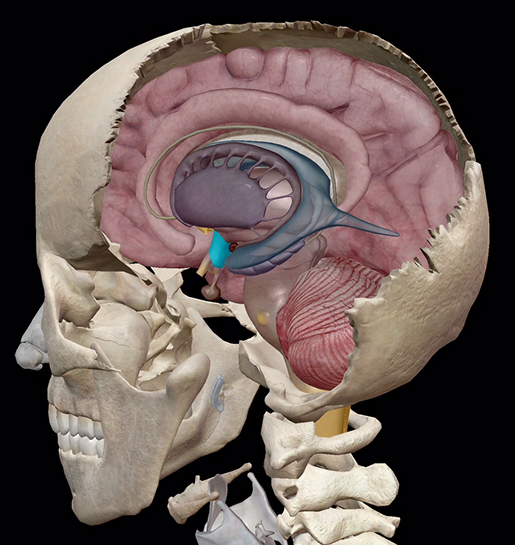 Image from Human Anatomy Atlas.
Image from Human Anatomy Atlas.
Since the hypothalamus is where it all begins, why not start here with it? Nestled below the thalamus and above the pituitary gland, the hypothalamus is a collection of specialized cells that serves as the central relay system between the nervous and endocrine systems. By secreting hormones that stimulate or inhibit production of other hormones in the anterior pituitary, this gland plays a crucial role in the release of hormones such as luteinizing hormone/follicle-stimulating hormone (LH/FSH—crucial for sex cell production), growth hormone–releasing hormone (GHRH—pretty self-explanatory), thyrotropin-releasing hormone (TRH) to regulate thyroid-stimulating hormone release, and corticotropin-releasing hormone (CRH) to regulate the adrenocorticotropin release vital to the production of cortisol (stress response hormone).
As you might imagine, in the instance of hypothalamic dysfunction or lack of function due to surgery, brain injury, tumors, etc., some of the aforementioned signaling processes may not occur. This can result in hormonal deficiencies with varying symptoms and severities. Luckily, treatment is often possible through hormone replacement therapies.
Pituitary
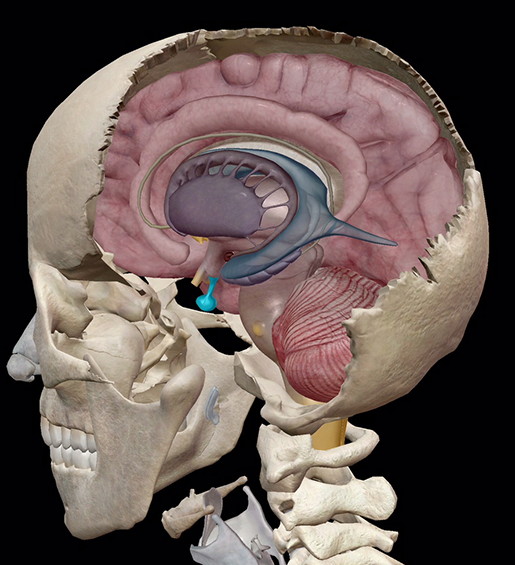 Image from Human Anatomy Atlas.
Image from Human Anatomy Atlas.
Whoever said bigger is better wasn’t too familiar with the pituitary. No bigger than a peanut, the pituitary gland is located at the base of the brain inferior to the hypothalamus, and is often considered the most important part of the endocrine system. It secretes endorphins (you know, the chemical root of happiness), as well as controlling several other endocrine glands, and regulating the ovulation and menstrual cycle. That is one busy gland.
Divided into two parts, the pituitary’s anterior lobe regulates the activity of the thyroid, adrenals, and reproductive glands by producing hormones that regulate bone and tissue growth in addition to playing a role in absorption of nutrients and minerals. This lobe is also responsible for the secretion of prolactin, a hormone vital to activating milk production in new mothers; thyrotropin, which stimulates the thyroid gland to produce thyroid hormones vital to metabolic regulation; and corticotropin, which is vital in stimulating the adrenal gland and the “fight-or-flight” response (don’t you adrenaline junkies fret, we’ll get back to this).
Not to be outdone by its neighbor, the posterior lobe releases antidiuretic hormone, which helps maintain the body’s water balance in addition to producing oxytocin, which triggers the labor contractions in the uterus.
Given its governing role in growth and development (and pretty much everything else), you can imagine that a malfunctioning pituitary isn’t good news for anyone, and you’d be right. An overactive pituitary or a tumor located on the pituitary can result in excessive production of all aforementioned hormonal signals. Excessive production of growth hormones, for example, will result in excessive growth of certain body parts, especially in young children. Treatment is usually medication-based or surgical in the case of a tumor. On the other hand, an underactive pituitary, which fails to produce adequate amounts of growth hormone, can stunt a child’s development, most noticeably height.
Thyroid
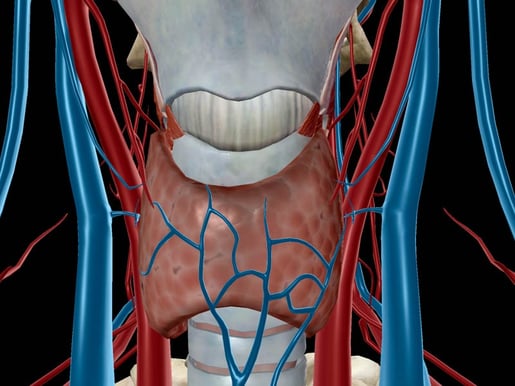 Image from Human Anatomy Atlas.
Image from Human Anatomy Atlas.
Encasing the trachea below the larynx sits the thyroid gland. Vital to growth and development, the thyroid hormone produced here regulates metabolism and nervous system activity, in addition to producing cells that secrete the hormone calcitonin, which aids in the regulation of blood calcium levels. While this gland might lack the laundry list of structures and functions of some others (we can’t all be the pituitary), it’s equally vital for the decisive role it plays in physical development by stimulating the growth and development of bone as well as brain and nervous system structures.
For all those fifty-somethings complaining about their sluggish metabolism, hyperthyroidism might seem like a dream. It’s not. In hyperthyroidism, the levels of thyroid hormones in the blood are excessively high; sufferers experience weight loss, nervousness, tremors, excessive sweating, increased heart rate and blood pressure, as well as protruding eyes and swelling in the neck from the enlarged gland. Inversely, in hypothyroidism, the levels of thyroid hormones are abnormally low, usually resulting in fatigue, slow heart rate, dry skin, weight gain, constipation, and, in children, slowing of growth and delays in puberty. In both cases treatment usually involves medications or surgery.
Parathyroids
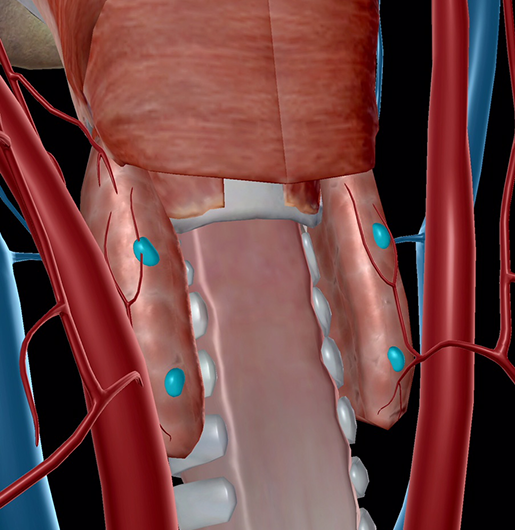
Image from Human Anatomy Atlas.
On the posterior surface of the thyroid sit the parathyroids. Tiny by comparison to the thyroid, these four glands secrete parathyroid hormone (PTH), which stimulates bones to release calcium into the blood when blood calcium levels are low. PTH also causes the kidneys to conserve calcium by reducing secretion into the urine in the instance of decreased blood calcium levels. Together, calcitonin and PTH act in complementary ways to maintain blood calcium homeostasis.
As you might imagine, too much PTH release (hyperparathyroidism), often caused by a tumor on the parathyroid gland, fosters a corrosive hormonal breakdown of bone, resulting in pain, fractures, and osteoporosis, in addition to heart palpitations and vascular blockages caused by excess calcium in the blood (released by the bones as they break down). Inversely, too little PTH release (hypoparathyroidism), which is often caused by accidental damage to the parathyroid during surgery or trauma to the head or neck, leads to abnormally low blood and bone calcium levels and an increase in blood phosphorus levels. While rare and often irreversible, side effects of these conditions are often treatable with supplements to manually manage bodily calcium availability and absorption.
Adrenal Glands
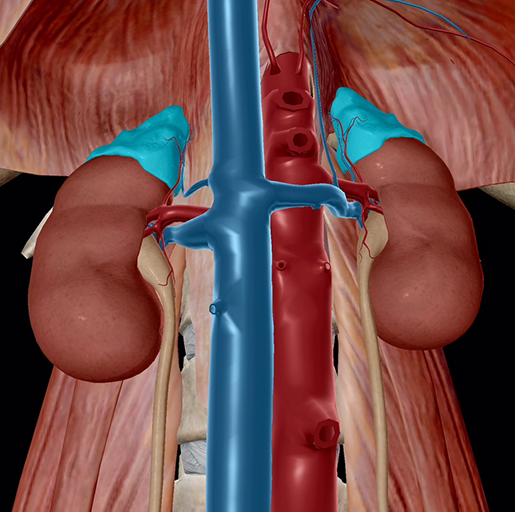
Image from Human Anatomy Atlas.
Pyramid-shaped organs perched atop each kidney, the adrenal glands consist of two structures: an outer adrenal cortex and an inner adrenal medulla. The adrenal cortex is a network of fine connective tissues that secretes a range of steroid hormones. These include glucocorticoids, such as cortisol, to manage protein and glucose levels; mineralocorticoids, which adjust levels of water and salt; and gonadocorticoids (aka androgens and estrogens). Last but certainly not least, the adrenal medulla produces epinephrine and norepinephrine (NE), chemicals vital to initiating the increased breathing and heart rate as the first step in the “fight-or-flight” bodily response to stress (told you we’d be back). These chemicals will also give your muscles a burst of energy, enabling you to either face that petrifying spider or run for dear life to find someone less arachnophobic to do so for you.
In small doses, the adrenal glands are crucial for arachnid confrontations as well as subsequent life-threatening encounters that you might wish to flee, yet overfunction of the adrenal glands doesn’t make you run faster, alas. Excess glucocorticoid hormone production can cause Cushing syndrome. Characterized by obesity, growth failure, muscle weakness, easy bruising of the skin, acne, high blood pressure, and psychological changes, the disease often results from a tumor or the use of synthetic corticosteroid drugs such as prednisone to treat autoimmune diseases such as lupus.
With decreased function of the adrenal cortex comes, no, not running slower, exactly, though you’re not far off. Underproduction of adrenal corticosteroid hormones often causes weakness, fatigue, abdominal pain, nausea, dehydration, and skin changes, and it’s typically treated, like many other hormonal deficits, by administering replacement hormones.
Pineal Gland
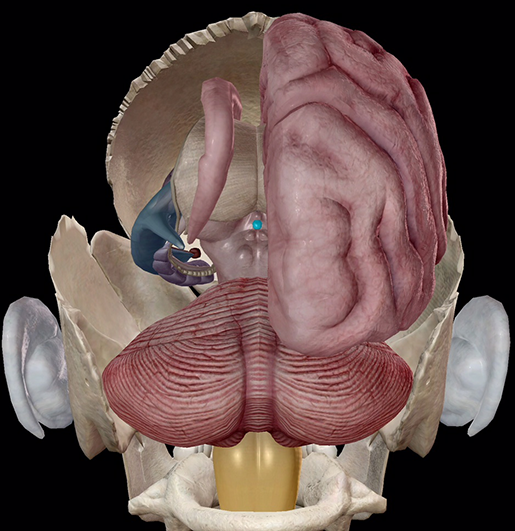 Image from Human Anatomy Atlas.
Image from Human Anatomy Atlas.
If you’ve ever traveled to northern Scandinavia or the Arctic Circle (typical summer destinations for the beach bums among us) and had your exotic polar holiday ruined by a lack of sleep, don’t blame your travel agent (or maybe do). It’s actually your pineal gland’s fault. Or maybe payback for all those all-nighters in college, who knows. This pinecone-shaped gland located at the posterior center of the brain activates with the absence of light at night to secrete the hormone melatonin, which in turn regulates our sleep patterns in both daily (circadian) and seasonal rhythms. In the morning (or at 1 a.m. north of a certain longitude in June), when light hits the eye, photoreceptors in the retina send signals to the pineal gland, which then decreases melatonin production, aka your body’s way of saying “Wakey wakey!”
Pancreas
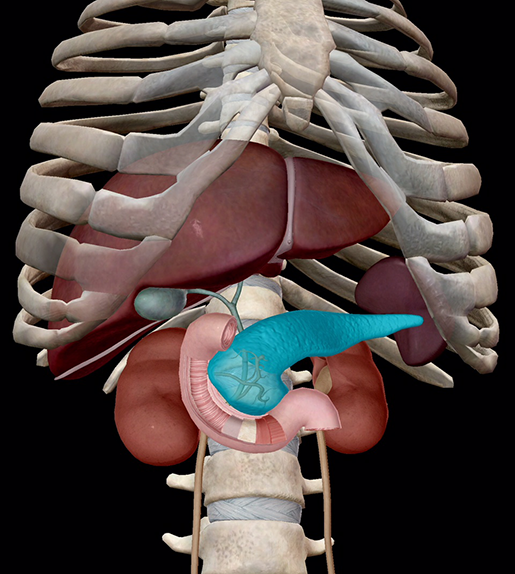
Images from Human Anatomy Atlas.
But before these glands get up on their high horses, it must be noted that other organs also secrete hormones. Despite their categorization as secondary organs of the endocrine system, their purpose is by no means subpar; in fact, these organs are working double time. A key facet of the digestive system, for instance, is the excretion of pancreatic juices into the small intestine via the pancreatic duct. The pancreas belongs to this special group of overachievers for its tiny cell clusters called pancreatic islets (or islets of Langerhans) that release hormones into the bloodstream. Despite making up less than 2% of pancreatic tissue, these islets regulate blood glucose levels (blood sugar) by signaling alpha cells in the islets to release glucagon when blood sugar is low, causing the liver to break down glycogen and release glucose into the blood, and beta cells when blood sugar is high to release insulin and glucose re-uptake.
If you need proof of this supposedly “secondary” organ’s primary importance within the endocrine system and beyond, just ask any diabetic. Broken down into types 1 and 2, diabetes is thought to be the result (in the instance of type 1) of an autoimmune disorder in which specific immune system cells and antibodies attack and destroy the insulin-producing cells of the pancreas. Rude. Symptoms include excessive thirst, hunger, urination (peeing), and weight loss.
The long-term complications of failing to treat type 1 diabetes with blood glucose monitoring and regular injections of insulin include kidney malfunction, nerve damage, blindness, and early coronary heart disease and stroke.
Unlike type 1, in which the body fails to produce normal amounts of insulin, in type 2 diabetes the body develops insulin resistance due largely to environmental conditions. Despite their differing causes, the symptoms and possible lethal complications of type 2 diabetes are largely the same as those of type 1.
Whether you’re stressed, pissed, thrilled, starving, or sprinting from danger, you likely owe how you feel (and how you deal with those feelings) largely to the endocrine system. So next time you feel like sneaking an extra donut or throwing a nasty tantrum, don’t be so quick to let yourself (or let your significant other) blame the hormones; they’re vital to how we enjoy those circular vessels of sugar and fat in the first place, and how we calm down post-conniption. And what’s a world without enjoyable donuts anyway?
Be sure to subscribe to the Visible Body Blog for more anatomy awesomeness!
Are you a professor (or know someone who is)? We have awesome visuals and resources for your anatomy and physiology course! Learn more here.
Additional Sources:
1. "Hormones and the Endocrine System." Hormones and the Endocrine System | Johns Hopkins Medicine Health Library. N.p., n.d. Web. 16 June 2017.
2. "Endocrine System." KidsHealth. Ed. Steven Dowshen. The Nemours Foundation, July 2015. Web. 16 June 2017.




